Orphan membrane transporters: mapping functions of the unknown
[[{“value”:”
Orphan membrane transporters: mapping functions of the unknown
Every cell in the body depends on transporters – proteins that move molecules in and out of the cell. While many are well understood, hundreds remain unknown. Dr Kathy Giacomini, Dean of the School of Pharmacy at the University of California San Francisco in the US, is investigating these orphan membrane transporters to reveal their roles in human evolution, normal physiology and the genetic reasons why people react differently to the same medicines.
Talk like a pharmacologist
Androsterone glucuronide — a natural waste product consisting of sex hormones that is filtered and sometimes reabsorbed by the kidney
Blood–brain barrier — a selective barrier of cells that prevents many substances from entering the brain from the blood, while allowing essential nutrients to enter
Confocal microscopy — a high-resolution imaging technique used to see the precise location of proteins within cells or tissues
Conjugate — a protein attached to a non-protein component, like carbohydrates or lipids
Deorphan — the process of discovering the function of an orphan gene or transporter whose role is unknown
Hominid — a member of the great ape family, which includes humans, chimpanzees, gorillas, orangutans, and their ancestors
Metabolite — substance produced through the chemical process of metabolism (which breaks down food, drugs or tissues for energy)
Orphan gene — a gene whose function is currently unknown
Solute Carrier (SLC) family — a large family of transport proteins that move a wide range of molecules across cell membranes
Stop-codon variant — a genetic change that causes the protein-coding sequence to stop prematurely, often producing a nonfunctional protein
Substrate — the specific molecule on which a functional protein acts
Zwitterion — a molecule that carries both a positive and a negative charge but is, overall, electrically neutral
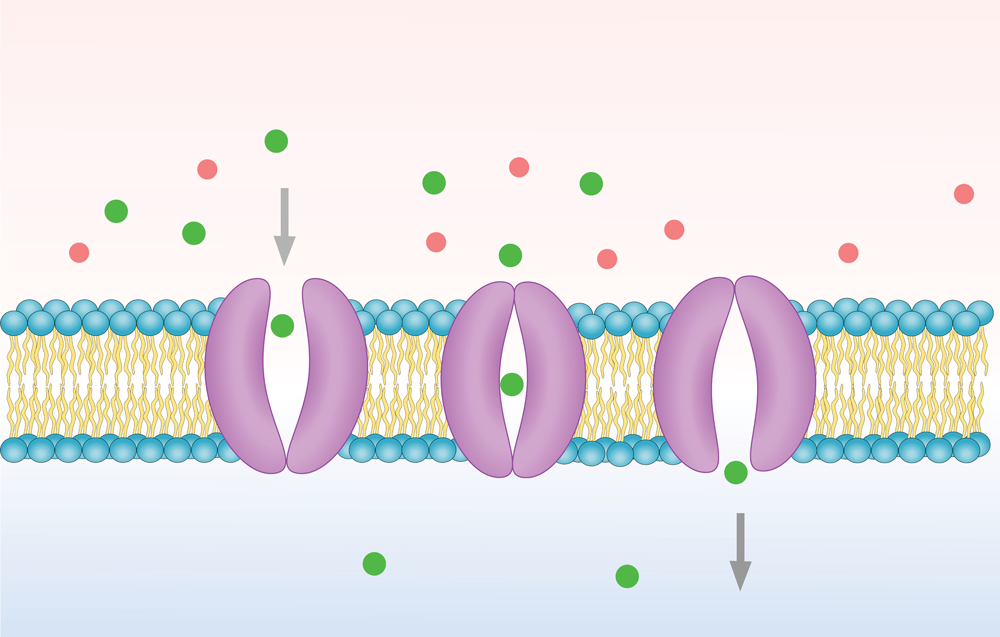
You might think that scientists already understand how every gene in our genome works. Surprisingly, around 20% of human genes are still orphans – meaning we do not know what they do. Some of these genes encode membrane transporters – proteins that move nutrients, drugs and waste products across cell membranes.
These transporters are vital for life. Without them, cells cannot absorb nutrients, remove toxins or respond to medicines. But for many transporters, their function remains unknown. “An orphan membrane transporter in the Solute Carrier (SLC) family is presumed to be a transporter, to transport molecules across the membrane, but its exact substrate or substrates have not been identified in humans or in any other species,” explains Dr Kathy Giacomini, Dean and Professor at the University of California San Francisco.
What is the Solute Carrier 22 family, and why study its orphan transporters?
“The transporters in the SLC22 family are fascinating because most of them are highly promiscuous, interacting and transporting a wide range of substrates,” says Kathy. Unlike highly specific transporters – such as the dopamine transporter, which handles almost exclusively dopamine – most SLC22 proteins can transport many different compounds. Because they recognise charge rather than a single defined molecule, they are known as the organic ion transporter family. Within this group are organic anion transporters, which move negatively charged molecules, and organic cation transporters (OCTs), which move positively charged ones. These transporters handle normal metabolites, environmental toxins, and many medicines, including penicillins and antiviral drugs.
How do scientists deorphan a transporter?
“The fun of deorphaning, the process of discovering a transporter’s function, is that there is no typical day,” says Kathy. Her team uses a wide range of techniques, from molecular biology to clinical studies. They may:
- tag a transporter with a fluorescent marker and use microscopy to see where it sits in the cell
- use metabolomic genome-wide association studies to see if genetic variants affect metabolite levels
- overexpress transporters in cells and measure which molecules are taken up more than in normal cells
- search public databases, including single-cell expression data, to find where transporters are present in the body
- analyse genome-wide association studies of traits or diseases to gain clues about a transporter’s role
- use confocal microscopy to locate transporters in specific tissues or in subcellular structures with high precision
- study the evolution of transporters across species, including ancient humans and other hominids.
Kathy’s team has also carried out clinical studies to see how transporter variants affect the body. Using genetic analyses, they identified people who carry a stop-codon in the orphan transporter SLC22A24, which prevents the protein from forming normally. They then compared urine and blood samples from these individuals with samples from people who have a functional transporter. “By analysing these samples, we tested whether people with the stop-codon variant cannot reabsorb androsterone glucuronide in the kidney and, therefore, eliminate more of it in their urine,” explains Kathy.
What has Kathy’s team discovered so far?
Reference
https://doi.org/10.33424/FUTURUM667
Left to right: Osatohanmwen Jessica Enogieru (graduate student), Lawrence Lin (staff and former graduate student), Huy Ngo (postdoctoral fellow), Niklas Handin (visiting student), Bianca Vora (graduate student), Megan Koleske (graduate student), Sook Wah Yee (staff researcher), Kathy Giacomini (professor), Dina Buitrago Silva (graduate student), Mina Azimi (postdoctoral fellow), Xujia Zhou (graduate student), Ling Zou (postdoctoral fellow)
© Giacomini Lab
Shuanglian Li (graduate student), Tom Urban (graduate student), Cheryl Cropp (graduate student), Sook Wah Yee (postdoctoral fellow), Debbie Lin (graduate student), Jim Shima (graduate student), Alexandra Ianculescu (graduate student), Ligong Chen (postdoctoral fellow), Swati More (postdoctoral fellow)
© Giacomini Lab
One of the team’s first discoveries in the orphan transporter area was the function of SLC22A15. They found that it transports zwitterions and shows a strong preference for ergothioneine, a powerful antioxidant that humans must obtain from food. Before this, only one human transporter – OCTN1 – was known to move ergothioneine. Yet OCTN1 is not present at the blood-brain barrier, even though ergothioneine is found in the brain. “Because SLC22A15 is located there, it may explain how this compound enters brain tissue,” says Kathy.
Kathy has also deorphaned SLC22A24, a transporter that moves sex-steroid conjugates such as androsterone glucuronide. Kathy’s team found that this transporter sits in the kidney, where it normally reabsorbs these molecules so they can be converted back into hormones like testosterone. However, many humans carry a common stop-codon variant that disrupts its function, and other primates are also gradually losing this transporter through evolution.
Finally, Kathy’s lab discovered that SLC22A10 works in chimpanzees but not in humans. In humans and ancient hominids such as Neanderthals, a single amino acid change destabilised the protein, preventing it from reaching the cell surface. When Kathy’s team studied the chimp version, they found it transported sex-steroid conjugates, hinting at a biological role that humans have lost over time.
Why does Kathy’s research matter?
Kathy’s team will continue to explore the SLC22 family, focusing on how the kidney reabsorbs steroid conjugates and recycles them back into active compounds. They are investigating how evolution has shaped these transporters, including why some have been lost in humans, and how this affects biological pathways.
“Ultimately, these studies will increase our knowledge of the genetic basis underlying drug response and will contribute to advancing the era of personalised medicine,” says Kathy. “Furthermore, our studies will explain the genetic mechanisms of decreased drug response and, ultimately, contribute to improving drug design for safe and effective treatments of subgroups of patients who do not respond to standard treatments.”
 Dr Kathy Giacomini
Dr Kathy Giacomini
School of Pharmacy,
University of California San Francisco, USA
Field of research: Pharmacology
Research project: Deorphaning membrane transporters
About pharmacology
Pharmacology is the study of how drugs interact with the body, including how they are absorbed, transported, metabolised and eliminated. Pharmacology studies help us understand why medicines work for some people but not others and how the body handles substances from food, the environment, or the microbiome. “Deorphaning a transporter, knowing that your research led to new knowledge of biology, is extremely rewarding,” says Kathy. “How can we understand human biology if we do not know what each individual gene does? Through this work, I feel I have contributed to an understanding of biology and, in particular, human biology.”
A major focus in pharmacology is pharmacogenomics, which studies how genetic differences affect drug response. Kathy’s research highlights how transporter genes influence the effectiveness of common medicines. For example, metformin, the first-line treatment for type 2 diabetes, is taken up by transporters in the liver called OCT1, OCT2 and OCT3. Studies in 10,000 patients showed that response to metformin was linked to a variant in a glucose transporter, SLC2A2, which controls how sugar is moved in and out of liver cells – rather than the SLC22 transporters. Meanwhile, allopurinol, used to treat gout, is converted in the body to an active form called oxypurinol, which relies on a transporter called ABCG2. A common genetic variant in ABCG2 affects how well the drug works, influencing both oxypurinol and uric acid levels. “All of these studies have important implications for precision medicine,” says Kathy. “With genotyping, one may be able to dose the drugs more precisely or to prescribe different drugs.”
The next generation of pharmacologists will have exciting opportunities, especially with new technologies. Computational methods, AI and advanced laboratory tools will help researchers study transporters in greater detail. “Learning how transporters interact with other proteins and the pathways that they form will be the subject of the next generation of transporter biologists,” says Kathy. “New methods to study transporters will also be developed, providing more research opportunities.”
Pathway from school to pharmacology
In high school, build a strong foundation in the core science subjects of biology, chemistry, mathematics and physics.
“At university, take courses in biochemistry, physiology, pharmacology and genetics,” advises Kathy. “It is also useful to understand computational sciences, such as how to program, as well as statistics.”
Look out for outreach opportunities provided by universities near you. “For example, we have many outreach schemes for high school students in the San Francisco Bay Area, most notably the University of California San Francisco Science and Education Partnership,” says Kathy. Find out more.
Explore careers in pharmacology
“Join various societies such as the American Society for Pharmacology and Experimental Therapeutics, the International Society for the Study of Xenobiotics, or the British Pharmacological Society,” says Kathy. These organisations provide educational resources, career guidance, webinars, and networking opportunities for students and early-career researchers.
Q & A
Meet Kathy
 Who or what inspired you to become a pharmacologist?
Who or what inspired you to become a pharmacologist?
I was strongly influenced by my parents. My father was a geophysicist who loved science, and I gained a strong love of science from him. My mother was a Filipina who had a strong interest in health professions. I became a pharmacist but was drawn to the sciences that form the underpinnings of the profession. So, I went back to school to get my PhD in pharmacology.
What experiences have shaped your career? Have there been any eureka moments along the way?
People have helped me along the way. My parents, my PHD advisor Dr Gerhard Levy, my postdoc advisor Dr Terrence Blaschke, who was open to me exploring various avenues of research, and my peer mentors at UCSF: Ira Herskowitz, Neil Risch and David Agard. Of course, my supportive husband, John, and family have been critical.
A eureka moment was when I realised that I had to learn molecular biology and went on a sabbatical in Dr Heini Murer’s laboratory to learn expression cloning of transporters. And a second was when I knew I had to learn more genetics and worked in the laboratory of Dr Yusuke Nakamura at the University of Tokyo in Japan. These two sabbaticals helped me change direction and refine my focus.
How do you switch off from the pressures of your work?
Research is an oasis and a love of mine. If I get a moment free from my very busy schedule as dean, I find my way to reading some interesting research. I am also an avid gardener and love growing vegetables. I love to crochet, read, play pickle ball and ski. I spend time with my three grown kids and have five grandkids, with another on the way!
What are your proudest career achievements so far?
I’m proud of many achievements. These include: cloning the first human organic cation transporter and the first human transporter in the SLC22 family, cloning the first human purine nucleoside transporter, CNT2, and deorphaning three transporters in SLC22 family. I am proud to have led the International Transporter Consortium, which has made excellent recommendations on transporter-mediated drug-drug interaction studies to the FDA and other regulatory bodies. My large genome-wide association studies in pharmacogenomics have yielded highly reproducible results, which has been very rewarding. I am proud of the many students and trainees I have mentored, who have gone on to have stellar careers in academia, industry and in drug regulatory agencies. And by no means least, I am proud of being dean of the number one school of pharmacy in the world!
What are your aims for the future?
My research continues! I am learning about the transporters I have already deorphaned, and I am deorphaning others. I would also like to explore the biological pathways of renal reabsorption and recycling for drug conjugates.
Pharmacy has a rich educational remit yet tends to only train pharmacists. I would like to lead the expansion of education programmes in schools and colleges of pharmacy. With others, I have already developed a new AI and Computational Drug Discovery and Development Master’s Degree programme and would like to do more.
Kathy’s top tips
1. Don’t follow in anyone’s footsteps.
2. Explore, enjoy and indulge your curiosity.
3. Have fun!
Do you have a question for Kathy?
Write it in the comments box below and they will get back to you. (Remember, researchers are very busy people, so you may have to wait a few days.)

Read about pharmacology research into biomarkers to help treat disease:
futurumcareers.com/searching-for-enigmatic-biomarkers-to-address-a-devastating-degenerative-disease
The post Orphan membrane transporters: mapping functions of the unknown appeared first on Futurum.
“}]]